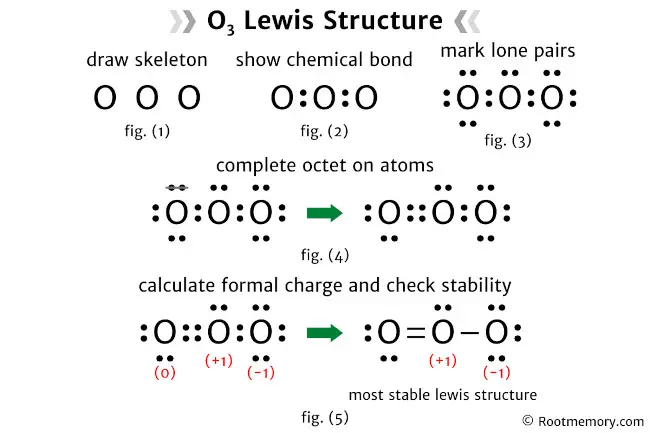
The Lewis structure of O3 illustrates the molecular arrangement of ozone, a molecule with three oxygen atoms. In this structure, there is one single bond and one double bond. The central oxygen atom forms a single bond with one oxygen atom and a double bond with the other oxygen atom. The oxygen atom bonded by the single bond has three lone pairs of electrons, the oxygen atom bonded by the double bond has two lone pairs, and the central oxygen atom has one lone pair. All the oxygen atoms satisfy the octet rule, and formal charges are present on two of the oxygen atoms. In the most stable Lewis structure of O3, the oxygen atom with the single bond has a negative (-1) formal charge, while the central oxygen atom has a positive (+1) formal charge.
Alternative method: O3 Lewis structure
Steps
Draw skeleton
In this step, first calculate the total number of valence electrons. And then, decide the central atom.
- Let’s calculate the total number of valence electrons
We know that… oxygen is a group 16 element. Hence, oxygen has six valence electrons.
Now O3 has three oxygen atoms.
So the total number of valence electrons = valence electrons of oxygen atom × 3
Therefore, the total number of valence electrons = 6 × 3 = 18
- Now decide the central atom
There are three oxygen atoms in O3. Hence, we can assume any one as the central atom. Let’s assume that center oxygen is the central atom.
So now, put three oxygens next to each other. And draw the rough skeleton structure for the Lewis structure of O3 something like this:
Also read: How to draw Lewis structure of H2S (4 steps)
Show chemical bond
Place two electrons between the atoms to show a chemical bond. Since center oxygen is surrounded by two other oxygens, use four electrons to show two chemical bonds as follows:
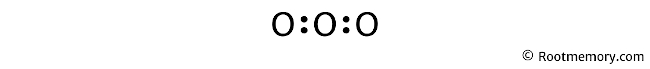
Mark lone pairs
As calculated earlier, we have a total of 18 valence electrons. And in the above structure, we have already used four valence electrons. Hence, fourteen valence electrons are remaining.
Two valence electrons represent one lone pair. So fourteen valence electrons = seven lone pairs.
Note that oxygen is a period 2 element, so it can not keep more than 8 electrons in its last shell.
Also, make sure that you start marking these lone pairs on outside atoms first. And then, on the central atom.
The outside atoms are left oxygen and right oxygen, so left oxygen and right oxygen will get three lone pairs. And the central atom (center oxygen) will get one lone pair.
So the Lewis structure of O3 looks something like this:
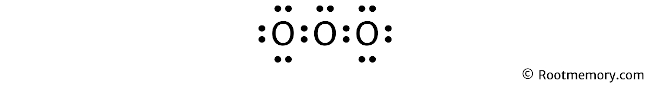
In the above structure, you can see that the octet is completed on outside atoms. But, the central atom (center oxygen) doesn’t form an octet.
So in the next step, we have to complete the octet on the central atom.
Also read: How to draw Lewis structure of C2H2 (5 steps)
Complete octet on central atom
Remember that oxygen is a period 2 element, so it can not keep more than 8 electrons in its last shell.
Now the center oxygen already has six valence electrons. Hence, the center oxygen needs two more valence electrons to complete its octet.
So convert one lone pair from the left oxygen atom to make a new bond with the center oxygen atom. And then, the Lewis structure of O3 looks something like this:

In the above structure, you can see that the octet is completed on the central atom (center oxygen), and also on the outside atoms. Therefore, the octet rule is satisfied.
After completing the octet, one last thing we need to do is, calculate the formal charge and check the stability of the above structure.
Also read: How to draw Lewis structure of SF4 (4 steps)
Calculate formal charge and check stability
The following formula is used to calculate the formal charges on atoms:
Formal charge = valence electrons – nonbonding electrons – ½ bonding electrons
Collect the data from the above structure and then, write it down below as follows:
- For left oxygen atom
Valence electrons = 6
Nonbonding electrons = 4
Bonding electrons = 4
Formal charge = 6 – 4 – ½ (4) = 0
- For center oxygen atom
Valence electrons = 6
Nonbonding electrons = 2
Bonding electrons = 6
Formal charge = 6 – 2 – ½ (6) = +1
- For right oxygen atom
Valence electrons = 6
Nonbonding electrons = 6
Bonding electrons = 2
Formal charge = 6 – 6 – ½ (2) = -1
Mention the formal charges of atoms on the structure. So the Lewis structure of O3 looks something like this:

In the above structure, you can see that the formal charges of atoms are closer to zero. Therefore, this is the most stable Lewis structure of O3.
And each horizontal line drawn in the above structure represents a pair of bonding valence electrons.
Next: Lewis structure of H2S
Related
- Lewis structure of H2S
- Lewis structure of C2H2
- Lewis structure of SF4
- Lewis structure of CCl4
- Lewis structure of OF2
External video
- O3 Lewis Structure – How to Draw the Dot Structure for O3 – YouTube • Wayne Breslyn
External links
- O3 Lewis Structure | Step By Step Drawing – What’s Insight
- O3 Lewis structure, Molecular geometry, Bond angle, Shape – Topblogtenz
- Ozone (O3) Lewis Structure | Steps of Drawing – Chemistry School
- O3 Lewis Structure in 6 Steps (With Images) – Pediabay
- O3 Lewis Structure, Polarity, Hybridization, Shape and Molecular Geometry – Geometry of Molecules
- What is the Lewis structure for ozone? – Quora
- O3 Lewis Structure, Molecular Geometry, Hybridization, and MO Diagram – Techiescientist
- Draw the Lewis structure for O3 – Homework.Study.com
- What is the Lewis structure of ozone (O3)? – Socratic
- O3 Lewis Structure – Laurence Lavelle
- Chemical Bonding: O3 Lewis Structure – The Geoexchange
- Can ozone have a triangular structure? – Chemistry Stack Exchange
- Lewis Dot of Ozone O3 – Kent’s Chemistry
- Draw the Lewis structure of ozone (O3) and then determine its molecular geometry – Numerade
- Why can’t the O3 structure be this? (see image) – Reddit
- Learn About Lewis Structure Of Ozone – Chegg
- Draw a Lewis diagram(s) for the ozone molecule (O3) – Vaia
- How many O3 resonance structures are there? [with study guide & diagrams] – AceOrganicChem
Deep
Rootmemory.com was founded by Deep Rana, who is a mechanical engineer by profession and a blogger by passion. He has a good conceptual knowledge on different educational topics and he provides the same on this website. He loves to learn something new everyday and believes that the best utilization of free time is developing a new skill.
